How Ginkgo biloba helps treat Alzheimer’s
Ginkgo biloba. Some of you might recognize it as a dietary supplement that is supposed to enhance cognitive function, but studies investigating these claims have mixed results. Xu Liu and colleagues recently investigated the effects of Ginkgo biloba extract on a mouse model for Alzheimer’s disease (AD). Interestingly, they were able to confirm positive effects on AD pathology, such as improved memory, but only after long-term treatment.
The cause of Alzheimer’s
One of the major hallmarks of AD is the build-up of amyloid beta (Aβ) plaques outside of the brain cells. These abnormal protein deposits cause inflammation, which damages the neurons, thus leading to the cognitive impairments seen in AD patients.
A natural extract
The dry extract from Ginkgo biloba leaves has exhibited anti-AD effects in several preclinical and clinical studies (for references, see the article from Liu et al.). To find out how this exactly works, Liu et al. focused on the effects of Ginkgo biloba on a molecular level.
Transgenic mice on supplements
The TgCRND8 APP-transgenic mouse overexpresses human mutated amyloid precursor protein (APP) in neurons, giving the mouse AD-like pathology. For Liu’s experiment, 2-month-old mice were given Ginkgo biloba extract as a dietary supplement for 5 months and 5-month-old mice were given the same dosage for 2 months. Control groups received the same diets, but without the supplement.
A long-term treatment
Some supplements and drugs have to be taken for a while before they start to have any effect. This seems to be the case for Ginkgo biloba as well: At the end of the test period, mice’s plasma levels were similar to levels found in humans taking the recommended dosage, inflammation in the brain was inhibited, and the loss of important proteins was attenuated. But this effect was far from immediate. Two months appeared to be too short a time frame to attain real results, and only the 5-month mice showed any significant improvement in cognitive function.
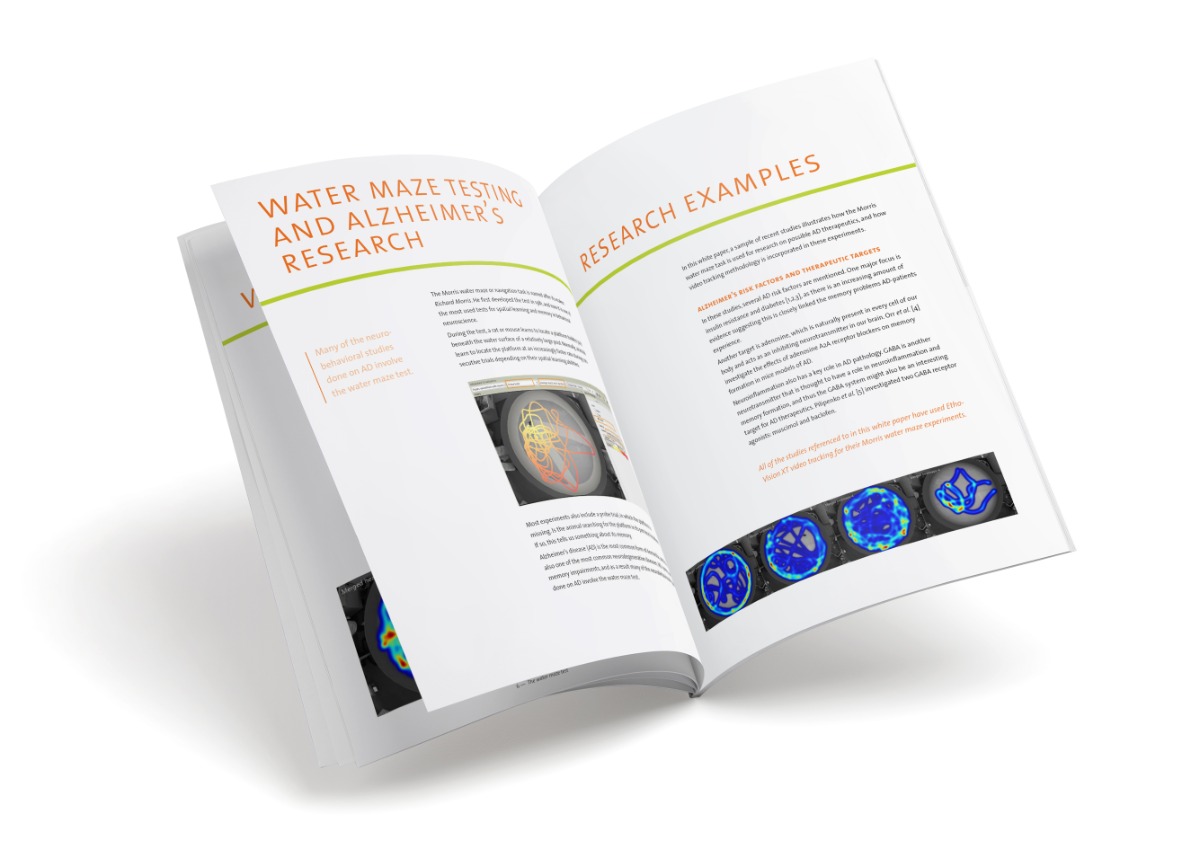
Remember where to go
The cognitive function of the mice was tested using a Barnes maze. This is a brightly lit, open disk with a number of holes around the circumference. One of these holes has a goal box underneath, providing a more preferable environment for the mouse. Mice typically learn to find this location using visual cues, similar to finding the platform in a Morris water maze test, and the traveling time and distance it takes them to enter the escape hole is used as a measure of spatial learning.

Ginkgo biloba helps in finding a safe place
Using video tracking (EthoVision XT), the traveling time, distance moved, and velocity of the mouse was automatically measured in the Barnes maze test. Researchers found that all the AD-mice were able to use the spatial reference points to learn the location of the escape box. The more training they got, the easier it was for them to find it, as was reflected by the decrease in distance moved and travelling time. They did not get there faster by moving faster; the velocity of the mice did not increase over trials. That said, the mice receiving Ginkgo biloba over 5 months showed a significant decrease in travelling time and distance, confirming the positive effects that were found on a molecular level in plasma and brain samples.
Ginkgo biloba helps in the long-term
This study shows that Ginkgo biloba extract treats the dementia symptoms of AD-mice through various mechanisms of protecting the brain cells and enhancing the concentration of important proteins in the brain, but it appears that a prolonged duration of supplementation is required. These results suggest that Ginkgo biloba may indeed be helpful in improving the symptoms and daily lives of Alzheimer’s patients.
Find out more
Read more in the complete study:
- Liu, X.; Hao, W.; Qin, Y.; Decker, Y.; Wang, X.; Burkart, M.; Schötz, K.; Menger, M.D.; Fassbender, K.; Liu, Y. (2015). Long-term treatment with Ginkgo biloba extract EGb761 improves symptoms and pathology in a transgenic mouse model of Alzheimer’s disease. Brain, Behavior, and Immunity, doi: 10.1016/j.bbi.2015.01.011.
- Read more about learning and memory tests in these blog posts, or about the application of the Barnes maze here.
Get the latest blog posts delivered to your inbox - every 15th of the month
more

How mice regain their memory: Betaine against Alzheimer’s Disease
As a potential Alzheimer medicine, betaine was tested on an AD animal model in a novel object recognition test. Using video tracking, scientists studied the influence of betaine and betaine transporter on mouse memory.
10 behavioral studies on Alzheimer’s and Parkinson’s
This blog post features 10 interesting studies that use innovative techniques to study models of AD and PD and important underlying neuronal mechanisms.