Dementia symptoms following surgery
This week we have a guest post by Iris Hovens. She has done some really interesting research into the consequences of surgery in terms of reduced memory and concentration problems. This is especially a concern for older people.
We are very happy that Iris has so kindly agreed to write about her research on our blog. At the end of this post, you will also find a link to a free white paper about this research! Thank you, Iris!
Characterizing postoperative cognitive dysfunction with a novel rat-model.
In the Netherlands, yearly more than 400.000 patients aged over 60 undergo surgery. Although the surgeries are aimed at improving health and well-being, ten percent of these older surgery patients will develop dementia-like symptoms, such as reduced memory and concentration and problems with planning and information processing.
This postoperative cognitive dysfunction (POCD) seriously affects the life of patients and their near friends and relatives, as it is associated with a reduced quality of life, increased dependency on social care and an increased risk of lasting mental and functional disability.
Translation in POCD research
The last decades many human and animal studies have been performed with the aim to understand POCD and its underlying mechanisms [1]. As the cognitive symptoms of POCD can vary widely among patients, human studies investigating the incidence and risk-factors have usually defined POCD as a persisting cognitive decline in general.
In contrast, mechanistic research in animals has focused on a simplified model of POCD investigating the effects of surgery on one specific brain region, the hippocampus, and the cognitive function related to this region, spatial learning and memory. These studies have greatly contributed to our understanding of POCD.
For example, we now have strong evidence that inflammatory processes that result from surgical trauma may play a key role in POCD development. However, the large differences in the view on POCD may hamper translation of animal studies into clinical practice.
A new rat-model for POCD
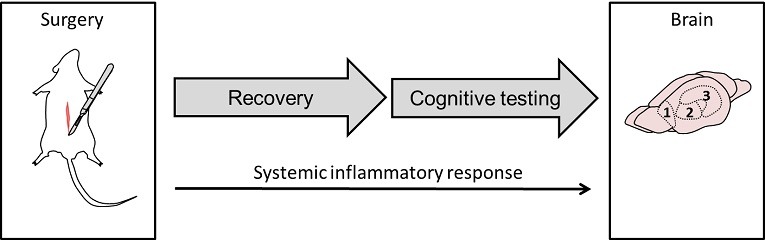
The aim of my PhD project was to develop a new model for POCD in rats to aid translation and gain more insight in the mechanisms underlying POCD. The model we developed made use of abdominal surgery in male rats, mimicking the effects of major abdominal surgery in humans.
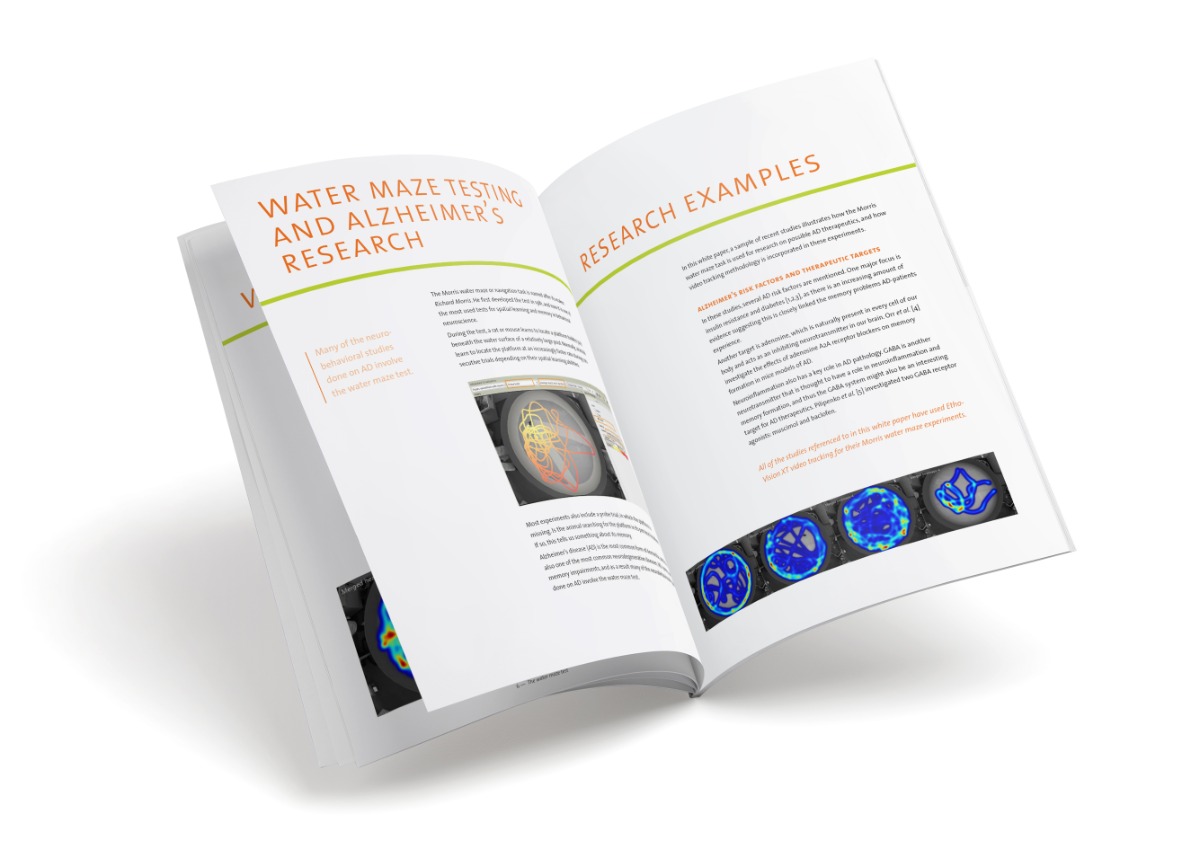
To be able to measure several aspects of cognition and mood related behavior in different time-windows of interest after surgery, we developed a compact behavioral test protocol (5 days, see figure). For this protocol we chose behavioral tests that could be performed quickly and of which the execution and analysis could be easily standardized using Ethovision software: An open field test to measure interest and anxiety, the novel object test and location recognition test to measure object recognition and spatial recognition, and the Morris water maze test to measure spatial learning, memory and cognitive flexibility.

Additionally, we analyzed markers for inflammation and neuronal functioning in the blood and in brain regions critically associated with cognitive performance: hippocampus for spatial learning and memory; prefrontal cortex for object memory; and striatum for cognitive flexibility [2].
Using this rat-model we investigated postoperative impairment in cognitive domains and related brain areas under conditions associated with different risk for POCD. As low POCD-risk condition we used healthy adult rats [2]. As elevated risk conditions we used aged rats (25 months) and adult rats that had experienced an infection prior to surgery [3,4].
Risk-factors determine the pattern of POCD
By researching multiple behavioral parameters and brain areas, we found that distinct cognitive domains and brain regions are differently affected by surgery. Spatial memory, regulated by the hippocampus, is particularly vulnerable to surgery and may even be impaired under conditions associated with low risk of POCD.
This finding promotes interpretation of postoperative cognitive complaints in low-risk patients. Conditions associated with an elevated risk for POCD are associated with more generalized postoperative cognitive and behavioral deficits. This is more alike to the situation in POCD patients and may therefore be a more representative and clinically relevant condition to study the underlying mechanisms of POCD.
Inflammation and POCD
We found that rats with an elevated risk for POCD show more extensive and prolonged neuroinflammation and changes in neuronal functioning in several brain regions critically associated with cognitive performance. This suggests that inflammatory processes may indeed be involved in the development of and vulnerability to postoperative cognitive impairment.
Further and future research
In addition to the experiments in our rat-model we investigate POCD in different cognitive domains in older surgical patients. The results of my PhD project will be defended at the University of Groningen on September 9, 2015. Future research will be aimed at the modulation of these inflammatory processes as a potential therapy for POCD.

References
- Hovens, I.B.; Schoemaker, R.G.; van der Zee, E.A.; Heineman, E.; Izaks, G.J.; Leeuwen, B.L. van (2012). Thinking through postoperative cognitive dysfunction: How to bridge the gap between clinical and pre-clinical perspectives. Brain, Behavior and Immunity, 26(7), 1169-79.
- Hovens, I.B.; Schoemaker, R.G.; van der Zee, E.A.; Absalom, A.R.; Heineman, E.; Leeuwen, B.L. van (2014). Postoperative cognitive dysfunction: Involvement of neuroinflammation and neuronal functioning. Brain, Behavior and Immunity, 38, 202-10.
- Hovens, I.B.; van Leeuwen, B.L.; Nyakas, C.; Heineman, E.; van der Zee, E.A.; Schoemaker, R.G. (2015). Prior infection exacerbates postoperative cognitive dysfunction in aged rats. American Journal of Physiology - Regulatory, Integrative and Comparative Physiology, 309(2), 148-159.
- Hovens, I.B.; van Leeuwen, B.L.; Nyakas, C.; Heineman, E.; van der Zee, E.A.; Schoemaker, R.G. (2015). Postoperative cognitive dysfunction and microglial activation in associated brain regions in old rats. Neurobiology of Learning and Memory, 118, 74-9.
Get the latest blog posts delivered to your inbox - every 15th of the month
more

New method for early detection of Alzheimer's disease
The color x-maze can be used to pick up symptoms of Alzheimer's disease in AD-mice months before traditional cognitive tests.
A 5-HT1A agonist and NMDA antagonist against Parkinson’s disease
Reducing side effects of L-Dopa with 5-HT1A receptor agonists can improve motor activity in a Parkinson’s disease model when combined with NMDA antagonists.